The "Organic Chemistry Nightmare"
For millions of NEET and JEE aspirants, from bustling coaching hubs in Delhi to students studying online, Organic Chemistry is a source of fear. It's seen as a gigantic, impossible mountain of 500+ "named reactions," endless reagents, and baffling exceptions that must be memorized by rote.
Students spend months creating flashcards, trying to cram every reaction into their short-term memory, only to find that it all vanishes in the exam hall. They can't solve multi-step synthesis problems ("Convert A to B"), and they panic when they see a "new" question that wasn't in the textbook. This is especially true when learning how to study organic chemistry for jee advanced, where problems are almost never straightforward.
Here is the single most important secret to this subject: Organic Chemistry is not a subject you memorize. It's a language you learn.
You wouldn't try to "memorize" the entire English language. You learn the alphabet (GOC), you learn the grammar (mechanisms), and then you practice forming sentences (solving problems). When you're fluent, you can create new sentences you've never spoken before.
This is how to master organic chemistry without memorizing. It's about shifting your entire mindset from "what" to "why." As a mentor who has guided thousands of students to top ranks, I can tell you that the moment this "click" happens, Organic Chemistry transforms from your most-feared subject to your highest-scoring one. These organic chemistry tips and tricks are all based on this one central idea: logic over rote.

A Note from Satyakam Sir
M.Sc. in Chemistry (Gold Medalist) | 15+ Years of Mentoring
"As an organic teacher with over 15 years of experience, my philosophy is that there are no 'exceptions' in Organic Chemistry, only deeper applications of fundamental logic. Stop memorizing, and let me teach you how to *think* like an electron. Once you see the logic, you can solve any problem."
The "factory model" of coaching, whether you find it in a large center or from a local chemistry tutor in Delhi, forces memorization because it's fast. But it's not effective. My entire teaching methodology is built on dismantling this fear. We will learn the "Why" behind every single reaction until it becomes second nature. Understanding organic chemistry reactions is the only path to success.
The 4-Step Method to Learn Organic Chemistry Logically
Forget the flashcards. This is the real pathway to mastery, from GOC for beginners to advanced synthesis.
Step 1: Master the 'Alphabet' — General Organic Chemistry (GOC)
The Common Problem: Students jump straight to "named reactions" (like Aldol or Grignard) without knowing *why* they happen. This is like trying to read a novel without knowing the alphabet. General Organic Chemistry (GOC) is the non-negotiable foundation.
You must have a crystal-clear, intuitive understanding of these four pillars:
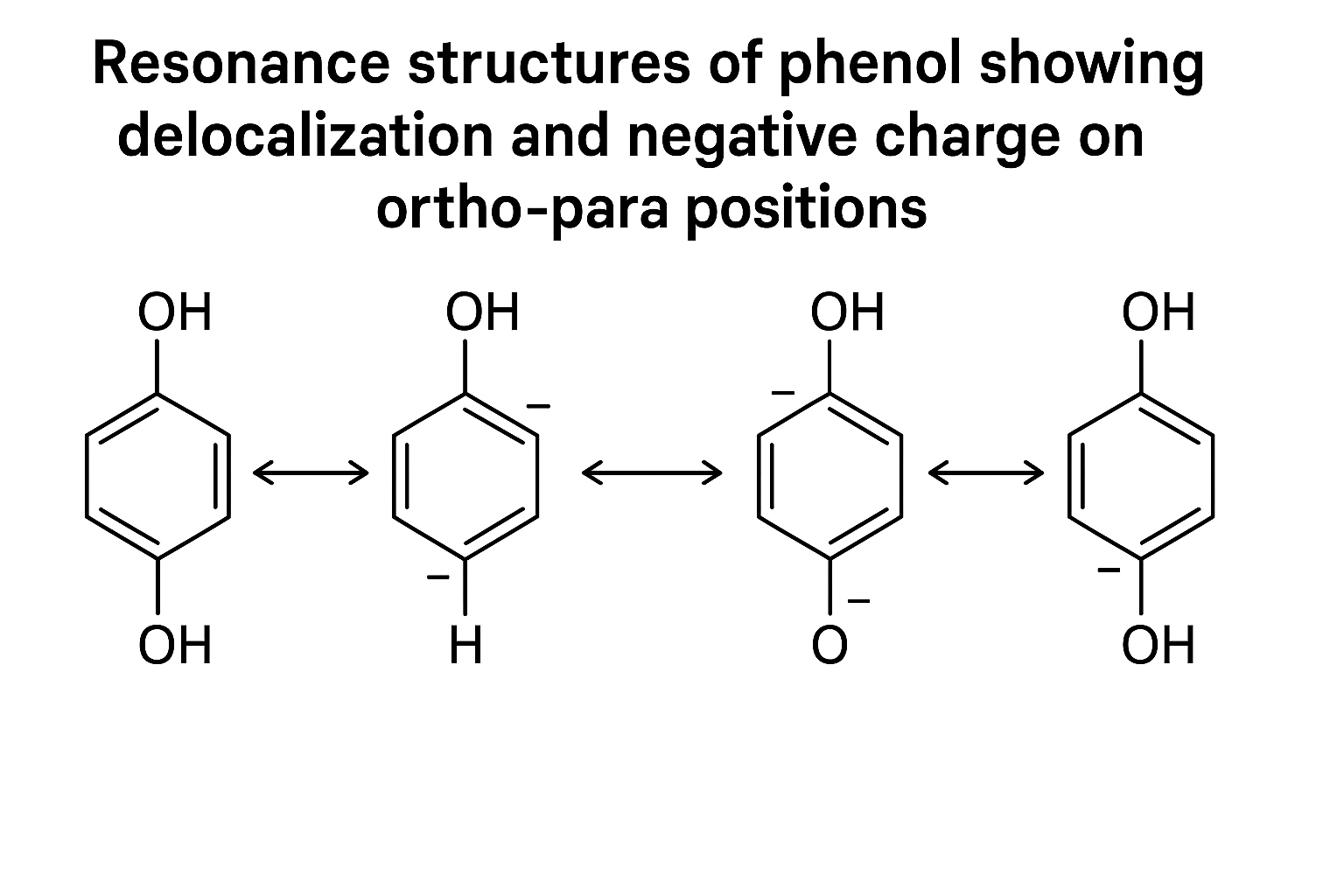
1. Resonance (or Mesomeric Effect)
This is how pi-electrons (in double bonds/lone pairs) move and delocalize. It explains stability and, crucially, *where* a molecule will react. A great mentor will teach you visualizing organic chemistry through this.
Look at the resonance structures of phenol. The lone pair on the oxygen atom delocalizes into the benzene ring. This does two things:
1. It makes the C-O bond stronger.
2. It puts negative charge density on the ortho and para positions (carbons 2, 4, and 6).
This is *why* phenol is an "ortho-para director" for electrophilic reactions. It's not a rule to memorize; it's a logical consequence of resonance.
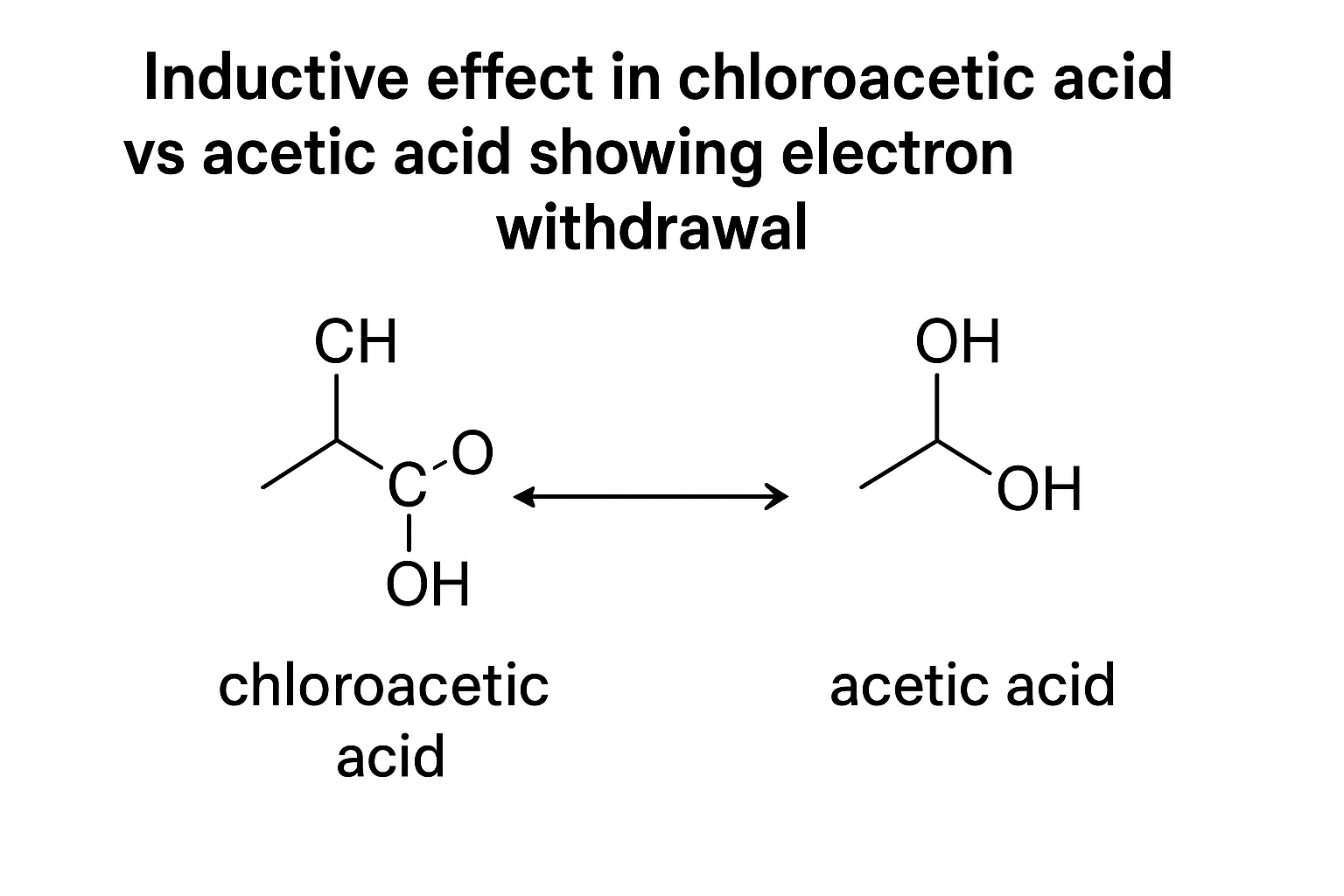
2. Inductive Effect
This is the "pull" or "push" of electrons through single (sigma) bonds due to differences in electronegativity. It's a weaker, permanent effect that explains acidity and basicity.
Why is Chloroacetic acid (Cl-CH₂COOH) stronger than Acetic acid (CH₃COOH)?
The highly electronegative Chlorine atom has a strong -I effect (electron-withdrawing). It "pulls" electron density away from the O-H bond, making the proton (H+) easier to remove. The resulting Chloroacetate ion is also stabilized. This is pure GOC logic.
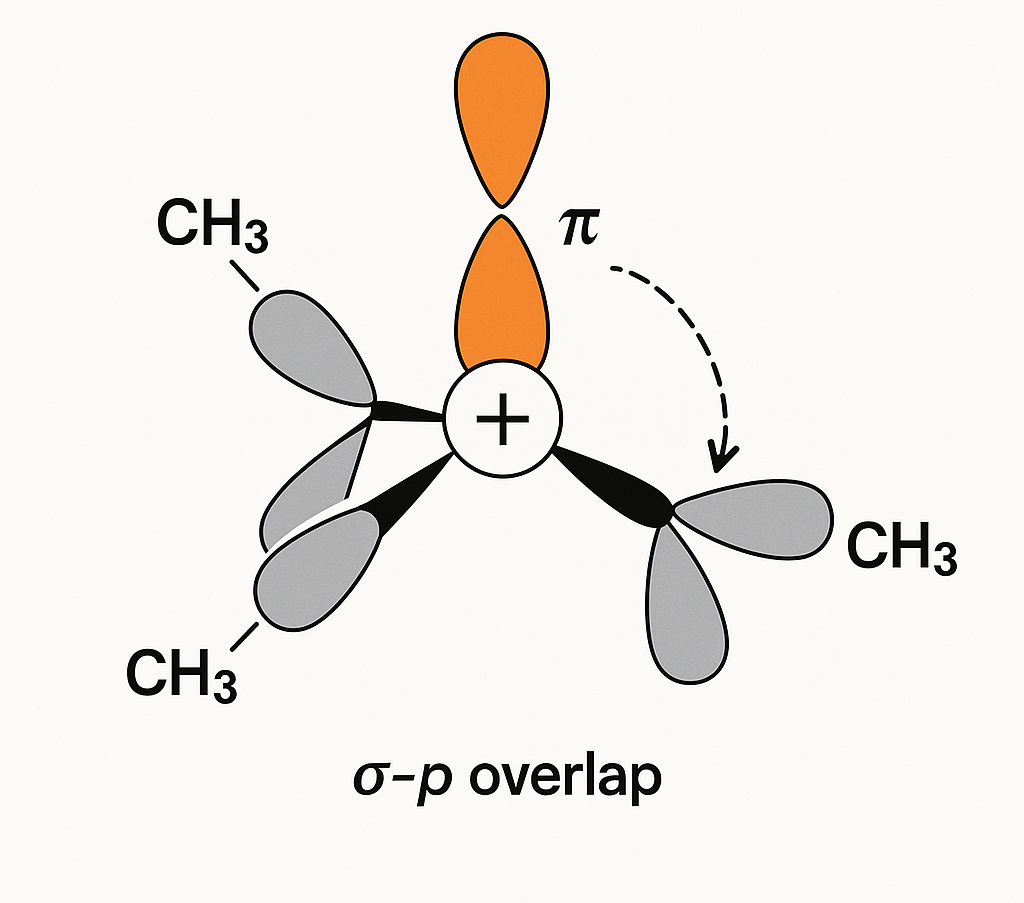
3. Hyperconjugation
This is the "secret weapon" for stabilizing carbocations and free radicals. It's the delocalization of sigma-bond (C-H) electrons into an adjacent empty p-orbital. The more C-H bonds (alpha-hydrogens) you have next to the charge, the more stable it is.
Why is a tertiary carbocation (like t-butyl) the most stable?
The central positive carbon is adjacent to *three* methyl groups, giving it 9 alpha-hydrogens. Each one can participate in hyperconjugation, spreading the positive charge. A primary carbocation has at most 3 alpha-hydrogens. More hyperconjugation = more stability. This dictates all SN1/E1 reaction pathways.
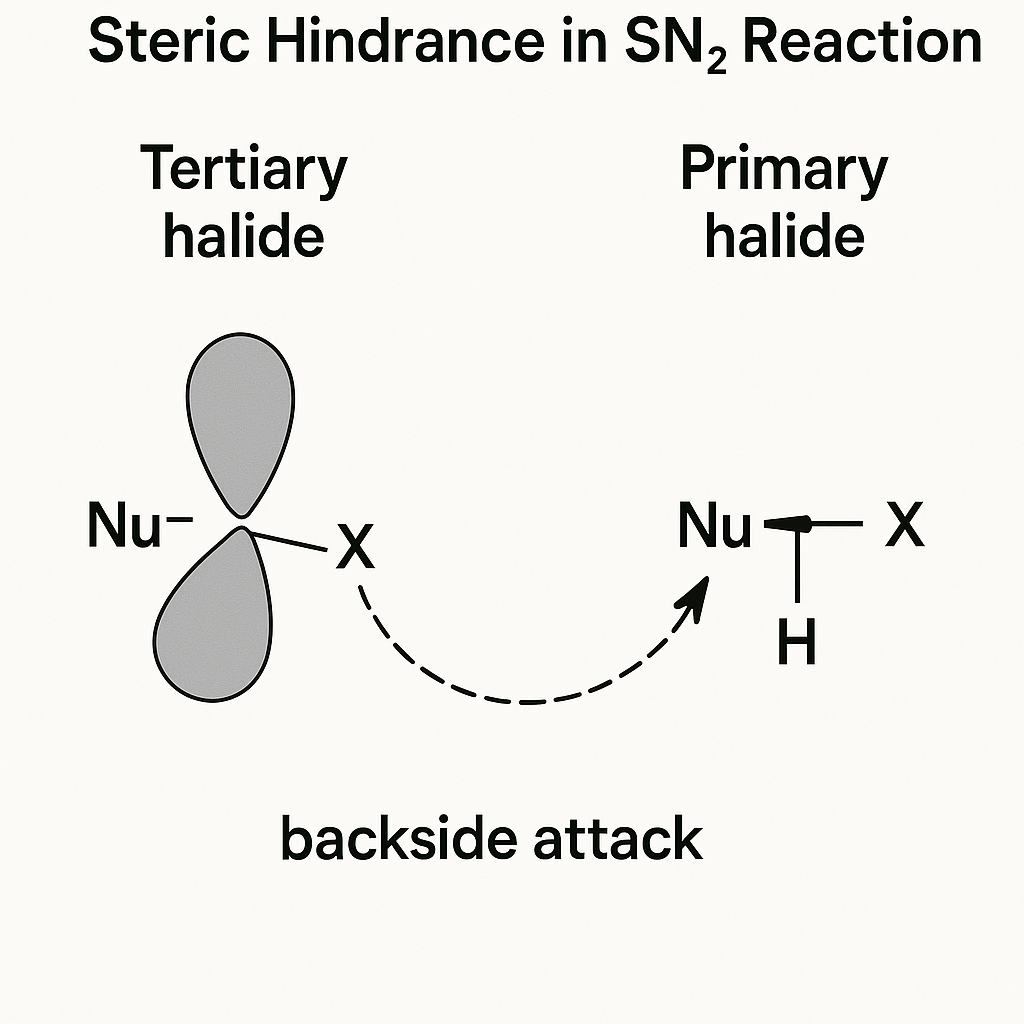
4. Steric Hindrance
This is simply the physical "crowding" of bulky groups. It's the "bouncer" of organic chemistry—it blocks reactions from happening at a specific site, even if it's electronically favorable.
Why does an SN2 (bimolecular) reaction fail on a tertiary halide (like t-butyl bromide)?
The SN2 mechanism requires the nucleophile to attack the carbon from the *backside*. In a t-butyl group, this "backside" is completely blocked by three bulky methyl groups. The bouncer (steric hindrance) won't let the nucleophile in. A primary halide (like methyl bromide) is wide open. This is a physical, logical concept, not a rule to memorize.
Step 2: Learn the 'Language' — Arrow-Pushing Mechanisms
The Common Problem: Students memorize: "Reagent A + Reagent B -> Product C." This is useless. When they see Reagent A + Reagent D, they are lost. The key is understanding organic chemistry reactions through their mechanisms.
A mechanism is the "language." The 'curved arrow' is its grammar. A curved arrow shows the movement of a pair of electrons. ALL organic reactions are just a flow of electrons from an electron-rich site (a Nucleophile) to an electron-poor site (an Electrophile). That's it.
Instead of memorizing 500 reactions, learn the 10-15 core mechanisms:
- Nucleophilic Substitution (SN1, SN2)
- Elimination (E1, E2)
- Electrophilic Addition (to alkenes/alkynes)
- Electrophilic Aromatic Substitution (EAS)
- Nucleophilic Acyl Substitution (in esters, amides)
- Nucleophilic Addition (to aldehydes/ketones)
- Radical Reactions
This reaction looks terrifying to memorize. But let's learn it using GOC logic:
[Image of Aldol condensation reaction mechanism step-by-step]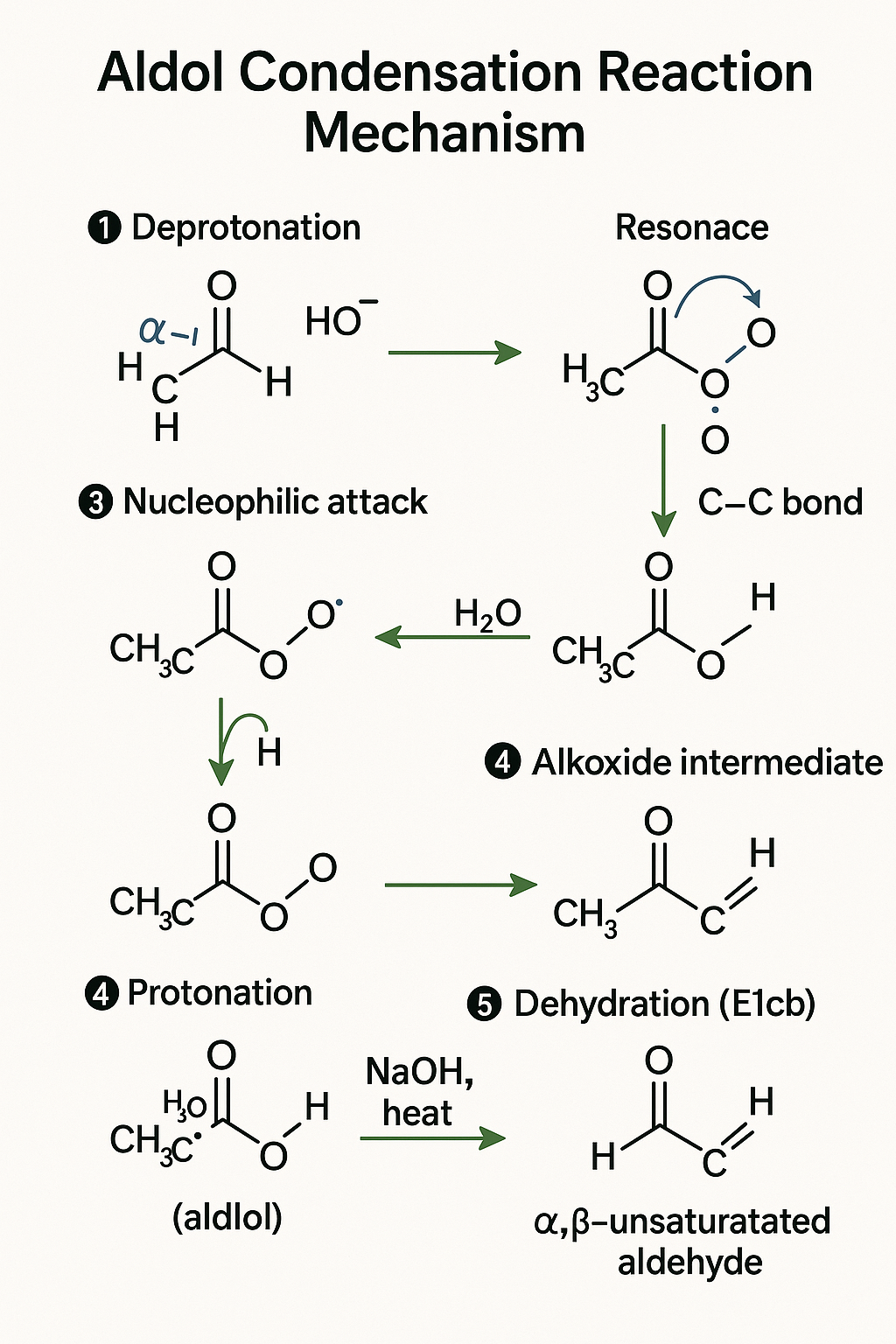
- Step 1 (Enolate Formation): A base (like OH-) is present. It needs a proton. Where is the most acidic proton? The alpha-hydrogen (on the carbon next to the C=O), because its conjugate base (the enolate) is stabilized by **Resonance** (GOC!). The base plucks it.
- Step 2 (Nucleophilic Attack): You have just created a strong nucleophile (the enolate). It "hunts" for an electrophile. The most electrophilic site is the carbonyl-carbon of *another* aldehyde molecule (due to the -I effect of the oxygen). The nucleophile attacks. A new C-C bond forms.
- Step 3 (Protonation): The resulting alkoxide (O-) is unstable. It takes a proton from water (which was formed in Step 1). You now have the "Aldol" (aldehyde + alcohol).
- Step 4 (Dehydration - *if heated*): The base plucks another alpha-hydrogen. The resulting carbanion kicks out the -OH group (a poor leaving group, but it works here) to form a new C=C bond. Why? Because the final product is an alpha-beta unsaturated carbonyl, which is an *extra stable conjugated system* (**Resonance** again!).
See? No memorization. Just a logical sequence of GOC principles. This is how to master organic chemistry mechanisms.
Step 3: Build 'Stories' — Reaction Roadmaps & Retrosynthesis
The Common Problem: Students can't solve "Convert Benzene to 2-phenyl-2-propanol." It looks impossible. This is a common challenge in how to study organic chemistry for jee advanced.
The Solution (Part A): Reaction Roadmaps
Don't think in a straight line; think in connections. Create visual "roadmaps" from key starting materials.
Start with "Propene" in the center. Draw "roads" to all the things you can make in one step:
→ (add H₂O/H⁺) → Propan-2-ol
→ (add HBr) → 2-Bromopropane
→ (add Br₂) → 1,2-Dibromopropane
→ (add O₃, then Zn/H₂O) → Ethanal + Methanal
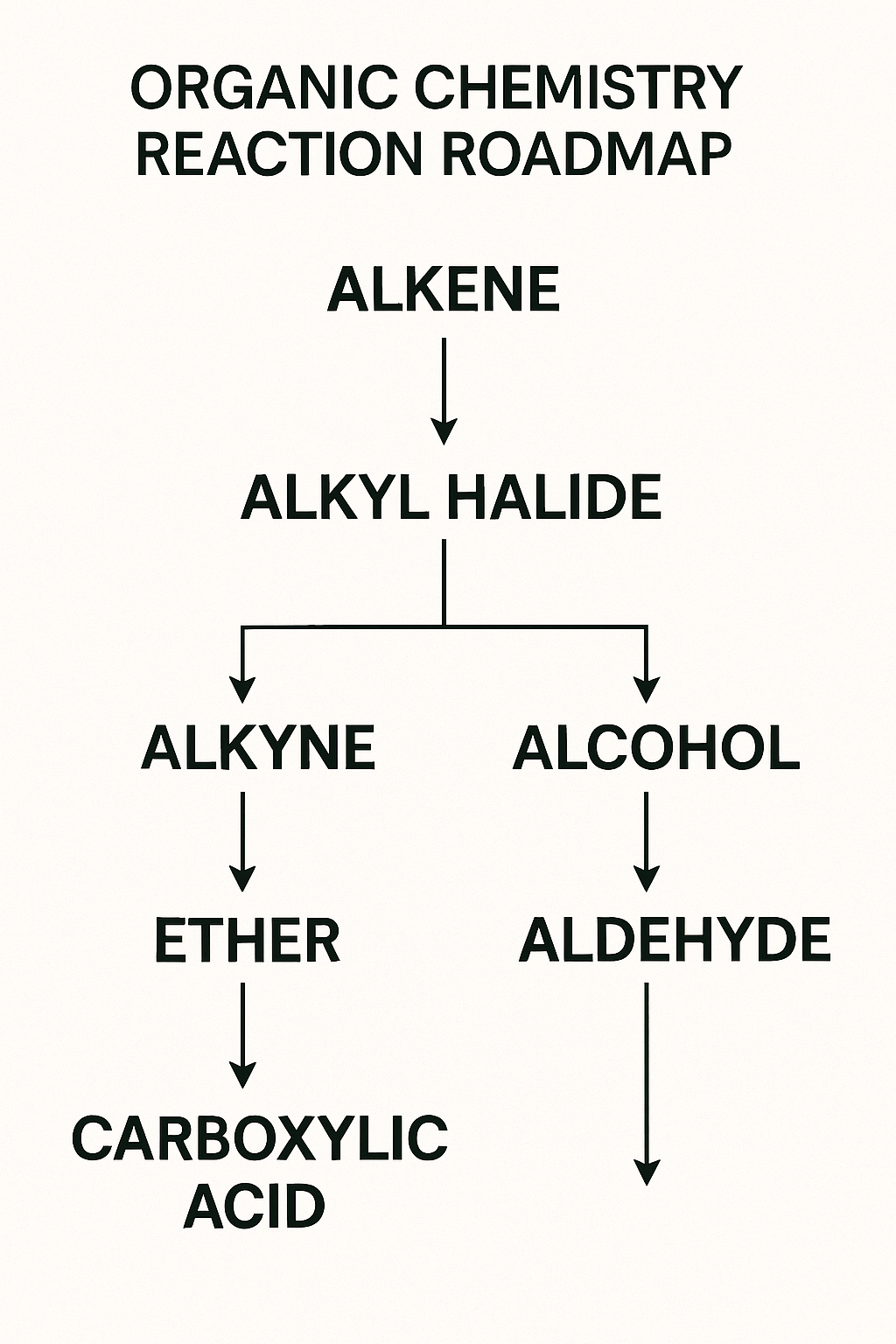
By building these maps, you create a visual library of your "tool-kit," making synthesis problems much easier.
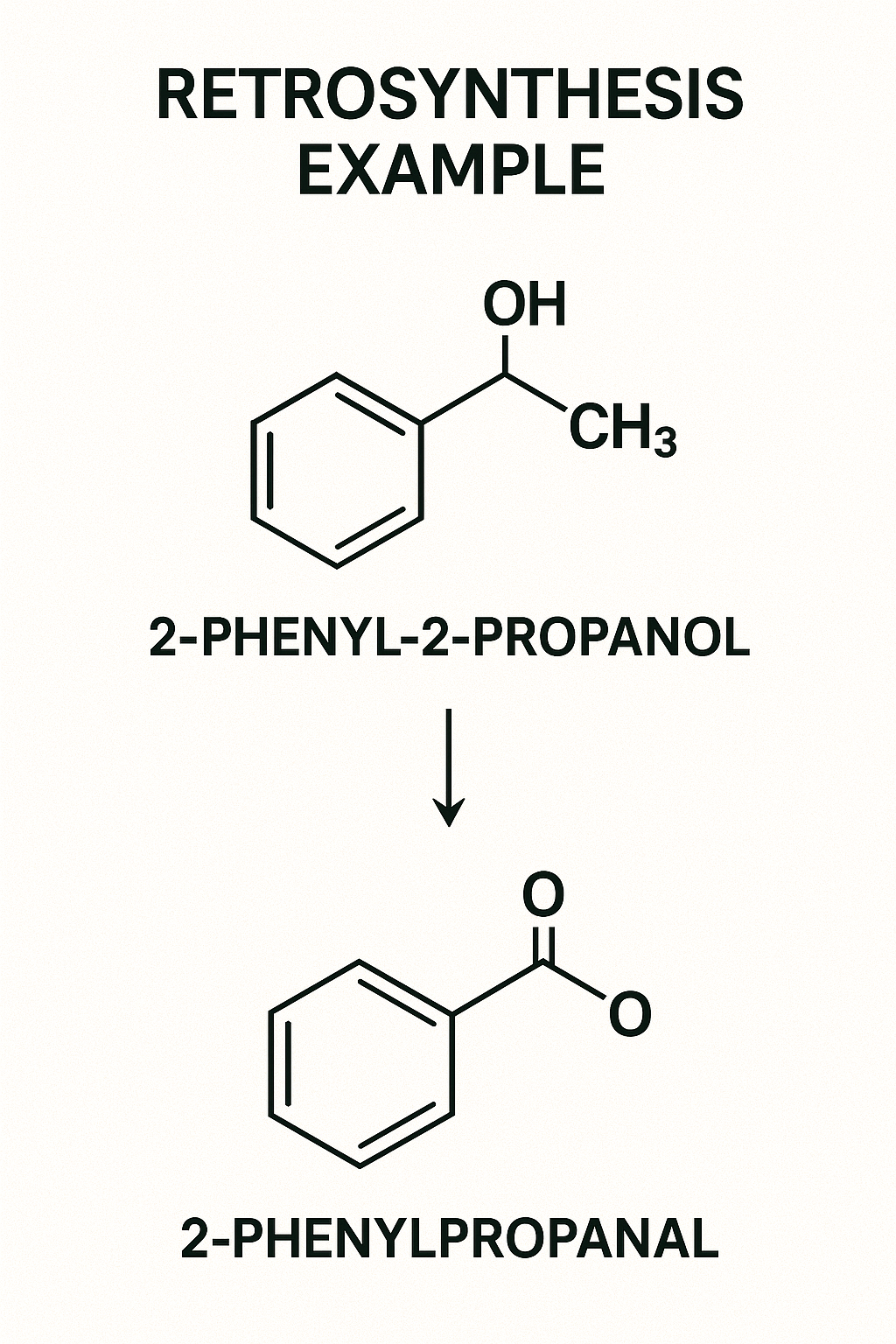
The Solution (Part B): Retrosynthesis (Work Backward)
This is the ultimate **organic chemistry tip and trick**. Don't look at the reactant; look at the *product* and ask, "How could I have made this?"
Goal: Synthesize 2-phenyl-2-propanol from Benzene.
1. Look at the Product: It's a tertiary alcohol. How do I make tertiary alcohols? A-ha! By reacting a **Grignard reagent** with a **ketone**.
2. Break it Down: I can make this from "Acetone" + "Phenyl Magnesium Bromide" (a Grignard).
3. Now, how do I make *those*?
- I need Phenyl Magnesium Bromide. I can make that from "Bromobenzene" + Mg.
- I need Bromobenzene. I can make that from "Benzene" + Br₂/FeBr₃ (EAS reaction).
4. Path Found!
Benzene → Bromobenzene → Phenyl Magnesium Bromide.
React that with Acetone, followed by H₃O⁺. Done.
You just solved a complex JEE Advanced problem by working backward, using logic, not memory.
Step 4: Achieve Fluency — Practice by Drawing, Not Just Solving
The Common Problem: Students read the textbook, watch a lecture, and solve MCQs. This is "passive" learning and "pattern recognition."
The Solution: You cannot learn a language by just reading the dictionary. You must *speak* it. You cannot learn Organic Chemistry by just *watching* it. You must *do* it.
This is the most critical step. Get a notebook and **draw out the full mechanism for every single problem** you do.
"Don't just solve MCQs. That's a test of your memory. Instead, take a blank page. Write down two reactants from a problem (e.g., Toluene + CH₃Cl/AlCl₃). Close the book. Now, from scratch, predict the product and *draw every single arrow* of the mechanism (the EAS mechanism, in this case). Talk to yourself: 'This is Friedel-Crafts. The AlCl₃ is a Lewis acid. It will pull off the Cl to make a CH₃⁺ electrophile...' and so on."
This "active recall" and "muscle memory" of drawing mechanisms builds deep, permanent understanding. This is the **best way to learn organic chemistry** for NEET and JEE.
🧪 Dynamic Experiments: Test Your Logic
Reading is one thing. Doing is another. Let's test your understanding of the GOC principles. Try to predict the outcome *before* you click to reveal the answer.
The Challenge: You have three molecules: Acetic acid (CH₃COOH), Chloroacetic acid (ClCH₂COOH), and Trichloroacetic acid (Cl₃CCOOH).
Your Task: Rank them from least acidic to most acidic.
Hint: Think about the Inductive Effect (-I). How does the number of electron-withdrawing Chlorine atoms affect the stability of the conjugate base (the anion)?
Logical Explanation:
Answer: Least Acidic: Acetic acid < Chloroacetic acid < Most Acidic: Trichloroacetic acid
The Logic: Acidity depends on the stability of the carboxylate anion (R-COO⁻) formed after losing a proton. A more stable anion means a stronger acid.
- Acetic acid: The CH₃ group has a weak *electron-donating* (+I) effect, which slightly *destabilizes* the negative charge by pushing more electrons onto the already negative anion.
- Chloroacetic acid: The single Cl has a strong *electron-withdrawing* (-I) effect. It 'pulls' the negative charge away from the oxygens, spreading it out (delocalizing) and *stabilizing* the anion.
- Trichloroacetic acid: Three Cl atoms have a *massive* -I effect. They pull electron density very strongly, spreading the negative charge significantly. This highly stabilizes the anion, making the parent acid very strong and eager to donate its proton.
You didn't memorize this; you *predicted* it with GOC!
The Challenge: You are running an SN2 reaction with Sodium Iodide (NaI).
Your Task: Which of these alkyl bromides will react the fastest?
- Methyl bromide (CH₃Br)
- Isopropyl bromide ((CH₃)₂CHBr)
- Tert-butyl bromide ((CH₃)₃CBr)
Hint: Remember the SN2 mechanism? The nucleophile (I⁻) must attack the carbon from the *backside*. Think about steric hindrance (the 'bouncer').
Logical Explanation:
Answer: Methyl bromide (CH₃Br) will react the fastest by a huge margin. Tert-butyl bromide will essentially not react at all via SN2.
The Logic: The SN2 reaction is all about access to the backside of the C-Br bond.
- Methyl bromide (Primary): The three tiny H atoms offer almost zero steric hindrance. The 'bouncer' is asleep, and the nucleophile can attack easily.
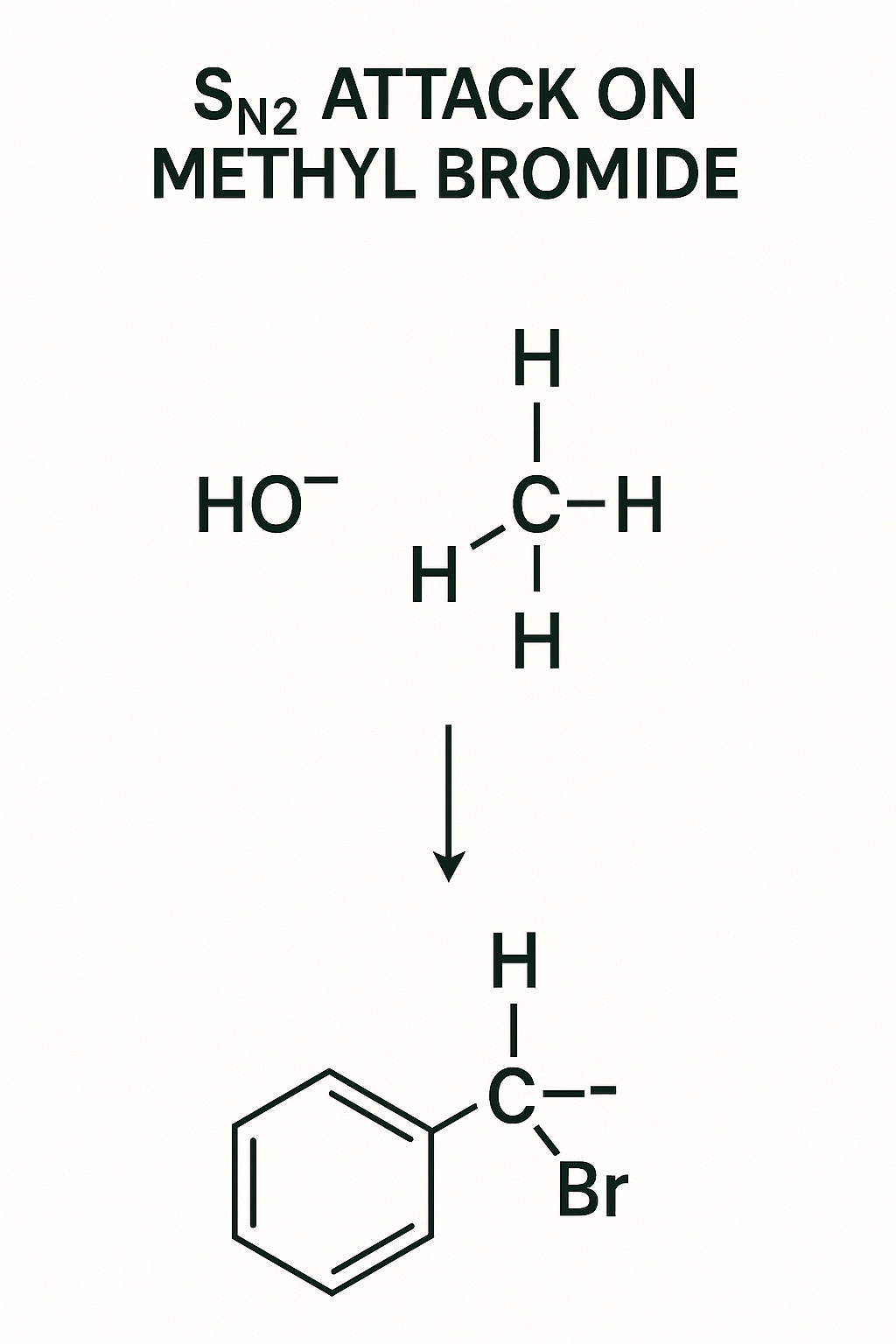
- Isopropyl bromide (Secondary): This is a secondary halide. The two CH₃ groups create some crowding. The 'bouncer' is awake and makes it difficult, so the reaction is much slower.
- Tert-butyl bromide (Tertiary): This is a tertiary halide. The three bulky CH₃ groups *completely block* the backside. The 'bouncer' has formed a wall. The SN2 reaction is "sterically forbidden."
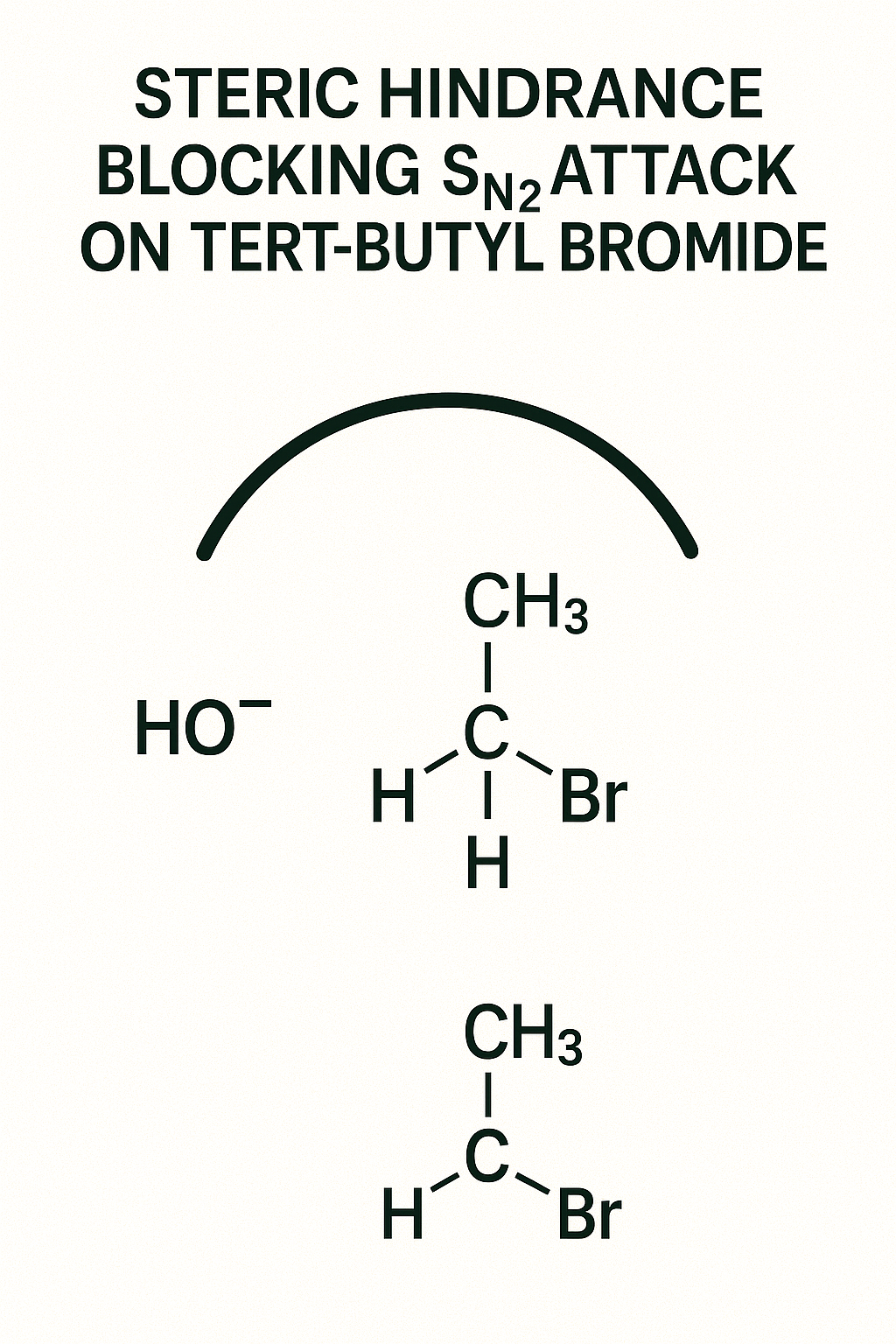
You just predicted a major rule of mechanisms based on simple physical logic!
The Challenge: You have three different carbocations.
Your Task: Rank them from least stable to most stable.
- Ethyl carbocation (CH₃CH₂⁺)
- Tert-butyl carbocation ((CH₃)₃C⁺)
- Allyl carbocation (CH₂=CH-CH₂⁺)
Hint: Stability comes from spreading out (delocalizing) the positive charge. What two GOC effects do this? Think Hyperconjugation (count α-H) and Resonance (pi-electrons).
Logical Explanation:
Answer: Least Stable: Ethyl carbocation < Tert-butyl carbocation < Most Stable: Allyl carbocation
The Logic: We analyze the stabilizing effects for each ion.
- Ethyl carbocation (Primary): This is a primary carbocation. It's only stabilized by Hyperconjugation from the 3 alpha-hydrogens on the adjacent CH₃ group. This is a weak effect, so this ion is very unstable.
- Tert-butyl carbocation (Tertiary): This is a tertiary carbocation. It has no resonance, but it has massive stabilization from **Hyperconjugation**—there are 9 alpha-hydrogens (from three CH₃ groups) to share the load. This makes it very stable.
- Allyl carbocation (Primary, but...): This *looks* like a primary carbocation, but it has a "secret weapon": Resonance. The positive charge is not stuck on one carbon. It is delocalized over *two* carbons via the adjacent pi-bond (CH₂=CH-CH₂⁺ ↔ ⁺CH₂-CH=CH₂). This resonance stabilization is a very powerful effect.
Generally, Resonance is a more powerful stabilizing effect than Hyperconjugation. Therefore, the resonance-stabilized allyl cation is even more stable than the hyperconjugation-stabilized t-butyl cation. You just used GOC to understand *why* these crucial intermediates form!

Stop Memorizing. Start Understanding.
If you're tired of feeling overwhelmed and are ready to finally *understand* Organic Chemistry, I am here to guide you. My entire teaching method is based on this logical, "first principles" approach. Whether you're looking for an organic teacher in Delhi or a mentor online, let's dismantle your fear and turn it into your greatest strength.
Book Your Free Demo Class Now!
